Introduction
With an increased emphasis on evidence based medicine, there has been an increasing focus on the pathophysiology of orthopaedic injuries and disease processes, and their impact on overall outcome. Traditional treatment strategies are evolving to encompass tailored approaches that account for age, occupation and patient expectation. Newer strategies for management are routinely implemented and encouraged in an effort to improve patient outcomes. It is to achieve these objectives that regenerative medicine which utilizes the use of stem cells and tissue engineering has newly emerged.
Definitions:
Stem Cells: Undeveloped biological cells capable of proliferation, self-renewal, conversion to differentiated cells and regenerating tissues1.
Totipotent cells: These are cells that can develop into all cell types in the human body and can also form extra embryonic and placental cells. The cells of the early stages of the embryo are the only totipotent cells and are not in clinical use due to ethical concerns2.
Pluripotent cells: Cells that can develop into cells of all the three germ layers (endoderm, ectoderm or mesoderm). Cells of late stages of embryo after the blastocyst stage are pluripotent cells2.
Multipotent cells: These are cells that can develop into more than one but not all germ layers. Adult stem cells and cord blood cells are multipotent cells2.
Types of Stem Cells:
Embryonic Stem Cells (Pre-natal): These cells are obtained from the blastocyst stage of the embryo. Pluripotent in the truest sense, they have a capacity to form into any tissue of the body and multiply in an unlimited manner. This is predominantly due to the phenomenon of asymmetric division – production of one stem and one non-stem daughter cell3. These properties, however, also make them prone to tumorigenesis4. This and the necessity of harvest from embryos causes safety and ethical dilemma.
Adult Stem cells (Post-natal): These cells are obtained later in life after the embryonic stage. They are multipotent, undifferentiated cells located among specialized tissues with a primary function of their maintenance and repair. Mesenchymal stem cells (MSC), which originate from the mesoderm, are a type of adult stem cells that have a good potential to develop into adipocytes, chondrocytes, myoblasts and osteoblasts.
Sources of Stem cells:
Stem cells can be obtained from bone marrow, periosteum, adipose tissue, placenta, umbilical cord, blood, human amniotic fluid, dental pulp, synovial tissue, skin and skeletal muscle. Among these, bone marrow, adipose tissue and muscle derived MSCs are most commonly used as they are easily obtained and abundantly available1.
Isolation of Stem cells:
Stem cells may be “unselected cells” obtained from autologous bone marrow after centrifugation or “selected” and enhanced in culture utilizing their affinity to tissue plastics. Cost involved, time to culture, risk of infection and loss of function in vitro are factors preventing regular clinical use of cultured MSCs. It should be noted that absolute number and the purity of cells obtained from cultures is higher, an important factor for clinical effect5. The posterior iliac crest has been shown to have a higher yield for MSCs as compared to anterior in case of bone marrow aspiration6.
Route of administration:
Stem cells may be directly applied into a lesion either surgically or via local injection with a suitable scaffold/carrier. MSCs may be taken through initial phases of differentiation, forming bone or cartilage precursors under laboratory conditions and then implanted into lesions. In addition, MSCs may be administered intravenously. Their ability to migrate systemically and colonize the bone marrow after a peripheral injection has been utilized for treatment of Osteogenesis Imperfecta7.
Mechanism of action:
In addition to differentiation into bone, muscle, cartilage, ligament or tendon cells, MSCs also have a paracrine effect whereby they secrete growth factors and cytokines such as bone morphogenic proteins (BMPs), transforming growth factor-β (TGF-β), and vascular endothelial growth factor (VEGF). These play an important role in angiogenesis, repair, cell survival and proliferation. MSCs also have the ability to migrate to the site tissue injury to modulate an inflammatory response8. Genetically modified MSCs for long term release of growth factors are being currently developed.
Role of Mesenchymal Stem Cells in Orthopaedic Surgery:
MSCs have an ability to develop into any mesodermal tissue. Thus, they can be prompted to form precursor cells to develop into tissues including bone, cartilage, muscle, tendon, and ligament. The use of stem cells for various orthopaedic challenges is outlined below.
Trauma and bone defects:
Nonunion/Delayed union and bone defects following trauma, tumor or infection are challenging aspects of orthopaedic surgery that may require biologic augmentation for optimum healing. Autologous cancellous graft is the current ‘gold standard’, but limited supply and donor site morbidity limit their use. Allografts and bone graft substitutes are routinely used to augment bone healing. However, poor graft integration and osteonecrosis of the graft remain primary issues with this method. Bone marrow aspirates that contain stem cells in a proportion of 1:10,000 to 1:1,000,000 of nucleated cells have been successfully used to enhance healing of non-unions5. Tissue engineering, involving the use of stem cells with scaffolds such as hydroxyapatite (HA), demineralized bone matrix (DBM) and tri-calcium phosphate (TCP), have been studied and found to be useful for bridging bone defects9. Due to absence of an extracellular matrix to grow on, MSCs alone have not proven to be beneficial for filling defects caused by simple/aneurysmal bone cysts10. Healing rates, are however, enhanced when these are used in conjunction with scaffolds.
Spine and peripheral nerve surgery:
Spine Fusion:
Neen et al, in a prospective study, showed that unselected stem cells used with HA scaffolds had similar healing rates as autologous grafting; thereby preventing donor site morbidity11. Similar results were obtained by Gan et al using β-TCP12.
Intervertebral Disc Degeneration:
Intervertebral disc degeneration is one of the most common causes of backache in a young productive population. Despite the high prevalence there is no treatment available which reverses the primary pathology. Animal experiments have shown increased proteoglycan content and maintenance of disc height with percutaneous stem cell injections13. Clinical trials are in progress to evaluate these results in humans with positive interim results14.
Spinal cord and peripheral nerve injuries:
Spinal cord and peripheral nerve injuries have a significant impact on quality of life of affected individuals. Animal studies have highlighted some positive effects of MSC use via intrathecal and local administration, however, the response seen in clinical studies is mixed15. In an animal study, Tamaki et al, demonstrated that muscle derived MSCs aided in successful regeneration of a crushed peripheral nerve16. Further prospective clinical studies are necessary to establish the role of MSCs in managing these patients.
Articular cartilage:
Articular cartilage is a highly specialized tissue with a poor intrinsic capacity to repair itself. The goal of any cartilage procedure is to restore its integrity so that it can withstand the wear and tear of daily activity.
Focal cartilage damage:
Since Pridie introduced subchondral drilling in the late 1950s, various procedures such as microfracture and abrasionplasty have been developed to recruit MSCs from adjacent bone marrow to proliferate into chondrocytes. Unfortunately, these procedures result in the formation of an inferior quality nonhyaline cartilage. Data on use of MSCs with suitable scaffolds in cartilage healing is mostly based on animal studies, with a few human case series showing improved healing and better function after autologous MSC implantation techniques17.
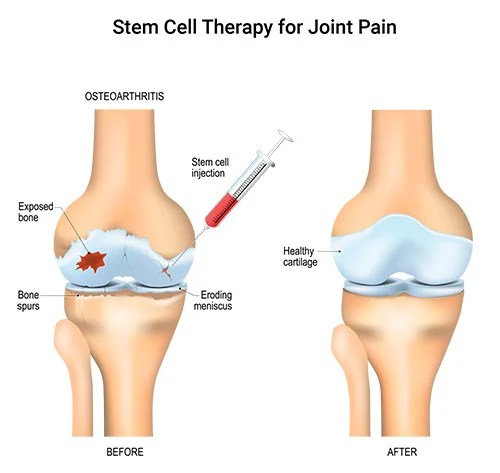
Osteoarthritis (OA):
Due to their role in inhibiting the catabolic activity of matrix metalloproteinases (MMP), MSCs have been shown to have a beneficial effect in OA18. In a recent study, Sato et al showed that guinea pigs with age related OA treated with MSC laden hyaluronic injections had better cartilage regeneration with higher type II collagen and lower MMP content19. Except for a few case series which show some clinical improvement there is a paucity of trials with human subjects which study the effect of MSCs on OA20.
High tibial Osteotomy (HTO) and Arthoplasty:
In a randomized control trial, Dallari et al showed that lyophilized bone chips treated as grafts with platelet gels and MSCs had higher rate of osteointegration in HTOs21. With appropriate use of nanotechnology to make optimum implant surfaces, MSCs have a great potential to revolutionize joint replacement surgery by facilitating osteointegration. Three dimensional scaffolds with MSCs may be used in the future to form autologous osteochondral grafts suitable for a ‘biologic’ arthroplasty22.
Avascular Necrosis:
Avascular Necrosis (AVN) of the head of femur is one of the most debilitating disorders in young patients. It is characterized by a decreased blood supply to the bone and associated increase in intraosseous pressure. The integrity of the subchondral plate is one of the most important deciding factor between head preserving (core decompression, bone grafting, femoral osteotomies) or head sacrificing (hip resurfacing/arthroplasty) procedures. Stem cells have angiogenic and osteogenic properties. Early stages of AVN are amenable to treatment with stem cell concentrate injection combined with routine retrograde procedures such as core decompression. Bone marrow aspirates administered after core decompression have been shown to be beneficial in AVN. Stem cells were isolated and used in a study by Rastogi et al where 60 hips in early stages of AVN were randomized to be treated either with core decompression and bone marrow injection or with core decompression and injection of isolated stem cells23. Two year follow up showed a better functional outcome and better radiographic healing in the stem cell group.
Wound Healing:
Although not typical in orthopaedic practice, poorly healing wounds are commonly encountered in treating patients with risk factors such as diabetes or open fractures. MSC treatment of acute and chronic wounds results in accelerated wound closure with increased epithelialization, granulation tissue formation and angiogenesis24.
Bone-Tendon interface and Tendon Healing:
Numerous commonly employed surgical procedures such as anterior cruciate ligament reconstruction, rotator cuff repair or retrocalcaneal bursa excisions depend on optimum healing of the bone-tendon interface. Fibrovascular scar formed during healing possesses inferior biochemical and mechanical properties. MSCs have been shown to promote early healing of the bone tendon interface by increasing the proportion of Sharpey’s fibers. MSCs used with bone morphogenic protein 2 (BMP-2) are associated with improved biomechanical properties of the bone tendon interface including stiffness and maximal load. A recent study by Adams et al showed that rats with Achilles tendon tear treated with stem cell-bearing sutures have higher failure strength and better histological properties25. Unselected MSCs were used for ultrasound-guided injections in a case series by Pascual-Garrido et al for chronic patellar tendinopathy with good clinical results26.
Paediatric Orthopaedics:
Osteogenesis Imperfecta (OI):
This is a heterogenous group of diseases with abnormality of type I collagen primarily leading to increased susceptibility to fractures, slow growth and loss of bone mass. Systemic infusion of allogenic MSCs by Horwitz et al in six children with OI showed improvement in bone mass and bone growth acceleration7.
Physeal injuries:
Bone bridge formation is an adverse complication following traumatic, infectious or other insult on the physis, leading to angular and/or longitudinal deformities. In a pig study, Planka et al showed that MSCs with scaffolds used in physeal defects differentiated into chondrocytes forming hyaline cartilage and prevented bony bridge formation27. Currently, there are no clinical studies to support this.
Osteoporosis:
In spite of tremendous advances in drug therapy, osteoporosis plays a significant role in overall health of geriatric patients. Increasing age is associated with decrease in number and function of osteoblasts and osteoprogenitor cells. Systemic infusion of MSCs has failed to promote bone formation due to their inability to migrate to the surface of the bone, a critical step for bone formation. In an animal study, Guan et al used MSCs modified to express certain surface proteins which enabled them to migrate to the periosteum leading to increased trabecular bone formation and bone mass28. Concepts such as these are positive steps towards utilizing MSCs for a generalized disease like osteoporosis.
Muscular dystrophies:
These are group of conditions wherein muscle fibers are replaced by fibrotic and adipose tissues, due to genetic mutations in several muscle proteins, which are essential for normal muscle function. There is no cure for these patients and treatment is focused on comfort care, respiratory assistance and delaying loss of function. Local and systemic transplantation of well differentiated myoblasts is associated with poor cell survival, limited migration from injection site and immune rejection. In a phase I clinical trial, Torrente et al showed that muscle derived stem cells with specific surface markers were safely transplanted in eight boys with no side effects29. Genetically modified MSCs are being developed for potential use of these cells in Muscular dystrophies.